Managing Vaccine Injection Site Reactions: Symptoms & Treatment
In this blog post, we will dive into the various types of vaccine injection site reactions, their symptoms, treatment and how to prevent and manage...
8 min read
Vaccine Injury Law Resources / Vaccines / Guide to Different Types of Vaccine Injections
 Paul Brazil
:
Nov 23, 2023 12:00:00 PM
Paul Brazil
:
Nov 23, 2023 12:00:00 PM
The proper administration of injections is critical in the healthcare field, and it’s beneficial to be aware of all their different types with distinct advantages/disadvantages and unique injection sites. This article discusses such details and stresses safety measures that should always accompany administering any injection.
Different injections serve different purposes: intradermal for skin tests, subcutaneous for vaccines, intramuscular for medications, and intravenous for quick action. The choice depends on the treatment's requirements. Vaccine injection site reactions refer to the body’s response to an immunization or other injection at the administration or injection site itself. These reactions can manifest as:
While some such injection site reactions may be mild and short-lived, others are more severe and require medical attention. Your safety and well-being are important, hence the necessity to recognize symptoms of a vaccine injection site reaction and understand when to seek medical help.
We shall explore the common and less common symptoms of vaccine injection site reactions and explore potential causes. We will also discuss the types of vaccine injection site reactions, including mild local reactions and severe reactions, and their respective symptoms.
When it comes to medical treatments, four key types of injections are used in healthcare settings: subcutaneous, intramuscular, intravenous, and intradermal. Each has its distinct purpose for use and must be administered properly depending on the patient’s condition or goal for care.
Both healthcare providers and patients must understand these distinctions so they can make informed decisions when it comes to medication delivery methods. This knowledge allows healthcare professionals to access a range of options according to specific needs while also ensuring safety standards within clinical procedures are upheld during treatment protocols.
In summary, understanding the different types of injectable drugs available - including subcutaneous, intramuscular, intravenous, or intradermal – provides us with an invaluable resource when it comes to selecting safe and effective injection solutions as part of wider health strategies moving forward
Subcutaneous injections commonly provide medication under the skin’s surface, in the fatty layer. They’re frequently employed for delivering insulin and other blood thinners like heparin into parts of the body tissue, either the lower abdomen or thighs. Regions with undulated or ridged texture should be avoided as this may result in penetration through muscle rather than subcutaneous tissue.
When getting ready for an injection, it is important to use one-time swabs designed specifically for clinical cleaning. Particularly those who suffer from diabetes can inject their medications using these methods.
An intramuscular injection delivers medications quickly into a person’s muscle tissue and can be employed for vaccines, antibiotics, or hormones. The best location for these shots is in the upper outer quadrant of the buttock area, with care taken to ensure proper injection technique so that there will not be any issues with nerve damage. To prevent leakage from occurring outside of the injected area when administering such an injection, healthcare workers commonly use what is known as ‘the Z-track method.' These injections may even need to be done at home by certain individuals themselves if necessary instead of relying on health professionals.
Intravenous injections are a way to quickly and effectively deliver medication directly into the bloodstream. Medical professionals must administer this type of injection due to their need for proper vein selection and technique, done on the elbow, arm, or hand area. When picking an IV catheter site, infections and joints where bending may occur must be avoided. While sore areas, stiff veins, or very thin ones should also not be chosen either. The selection of adequate veins is important to ensure safe yet effective administration through these injections.
Intradermal injections are distinct from a subcutaneous injection and intramuscular injection due to being delivered between the layers of skin. These kinds of injection techniques have specific purposes. Namely, allergy testing and immunizations such as TB skin tests.
While conducting an intradermal injection for allergy evaluation, it is important to be mindful when picking the area that will receive the shot – areas with moles or scars should not be selected since this may lead to incorrect test results, while locations affected by rashes or abundant hair growth must also usually be excluded. An exception can apply when a steroidal treatment through these types of shots needs to be administered (e.g., psoriasis).
Selecting proper injection sites is crucial for administering medications effectively and avoiding problems, including infection or undesired outcomes. The type of medication, patient conditions, and treatment objectives play a role in determining the best site. This article will investigate appropriate areas to inject different drugs and applicable methods.
To ensure proper absorption of the medication and reduce pain or discomfort during the injection, an intramuscular injection is most often given in larger muscle groups such as the deltoid (in the upper arm), vastus lateralis (thigh), and anterolateral aspect of the thigh. Medical professionals must take several steps before administering this kind of injection. These steps include confirming the patient’s identity, reviewing information on the medicine used, checking the prescription chart, and removing air from the needle before finally cross-verifying accuracy with orders mentioned in the administration record.
When giving subcutaneous injections, choosing the right injection site is important. Areas with sufficient fat - such as arms, abdomen, and legs - are most suitable for this procedure. Areas that appear sunken or lumpy should be avoided so the needle doesn’t penetrate muscle instead of subcutaneous soft tissue only.
Before administering any type of injection into a person’s skin, you must ensure the site is disinfected using a sterile swab specifically designed for medical hygiene.
The safety of patients and the successful administration of intravenous injections depend upon the proper selection of veins for this purpose. Healthcare professionals must assess vein accessibility, flow rates, and their patient’s current state to choose correctly.
It is typically accepted that the arms—both upper and lower—present low risks while being easily available for such interventions. To help these treatments, hydration should be provided as well as warmth. Applying a topical black pepper also provides improved visibility when necessary during injection processes. Aseptic techniques are essential, so medical personnel should seek expert advice if questions arise regarding them or other aspects of the IV injection administrations.
When determining the best type of injection for any given medical need, it is important to consider each method's advantages and disadvantages. This segment will analyze the plusses and minuses of subcutaneous injections, intramuscular shots, and intravenous injections to aid in making an informed decision on which approach would be ideal for your situation.
Subcutaneous injections are typically used for administering insulin, blood thinners, and other drugs that must be given in a low dose. This injection type provides advantages over alternative approaches, such as lower pain levels and the ability to self-administer or receive assistance from healthcare providers with special drug delivery systems. Studies have found subcutaneous shots may save money compared to intravenous dosing since systemic side effects are less likely.
Subcutaneous injections can cause lumps or nodules to appear at the injection site due to lipohypertrophy, a thickening of the subcutaneous fat layer around blood vessels, and inflammation and trauma caused by repeated injections. When rapid medication delivery is needed, these results may be slower than with intramuscular shots. For instance, the insulin absorption rate through an intramuscular shot is twice that of a subcutaneously injected form.
An intramuscular injection is a widely utilized method for delivering medication, particularly vaccinations. This is due to their rapid absorption rate and rich blood supply in deep muscle tissue, providing increased local blood flow at the injection site when administered properly.
Often referred to as an IM injection in medical circles, intramuscular injections are a common method for delivering medications. They are particularly useful for vaccines, allowing the medicine to reach deeper layers and promote a more effective immune response. This differs from IV or intravenous injections, which are unsuitable for vaccines. Intramuscular injections work quickly, as the medicine is absorbed rapidly from the muscle area where it's injected. Trained medical professionals typically handle this method of administration.
Subcutaneous injections are preferred over intramuscular ones due to less pain and discomfort. This is because a larger needle size is used for the latter, as it needs to reach deeper into muscle tissue containing more nerve endings. Care should be taken regarding proper injection site selection when dealing with an intramuscular shot since incorrect sites can lead to long-term effects such as lingering pain at that spot or even abscess formation or gangrene in some cases. Alternatives like joint injections may also be employed instead of regular IM shots. All this goes on top of needing carefully considered use whenever administering these types of needles and injections
Certain measures must be taken to ensure safety when administering injections and reduce the potential for complications like infection, adverse reactions, or errors in administration. To this end, proper hand hygiene habits must be followed along with sterile techniques when preparing medication and monitoring during injection delivery. These steps will be covered in detail in upcoming sections so everyone understands how best to perform injections safely.
The World Health Organization recommends washing hands and using clean techniques when preparing an injection to avoid infection. This means washing hands with soap and water or using hand sanitizers to eliminate bacteria. It's also important to use new needles and syringes and to keep the preparation area clean. These steps help ensure injections are safe and reduce the risk of infection.
To ensure safe and effective injection administration, it is necessary to take proper care in injectable medications' preparation, storage, and dosage verification. Such medicines should be kept according to their recommended conditions. Some might need refrigeration or freezing, while others must remain at room temperature (59-86 degrees Fahrenheit/15-30 Celsius).
During medication setup for injections, such measures as aseptic techniques with single-use needles and sterile syringes are important factors that help prevent contamination from occurring on equipment used and drugs administered. Observing hand hygiene before any preparations minimizes possible infection transmission risk.
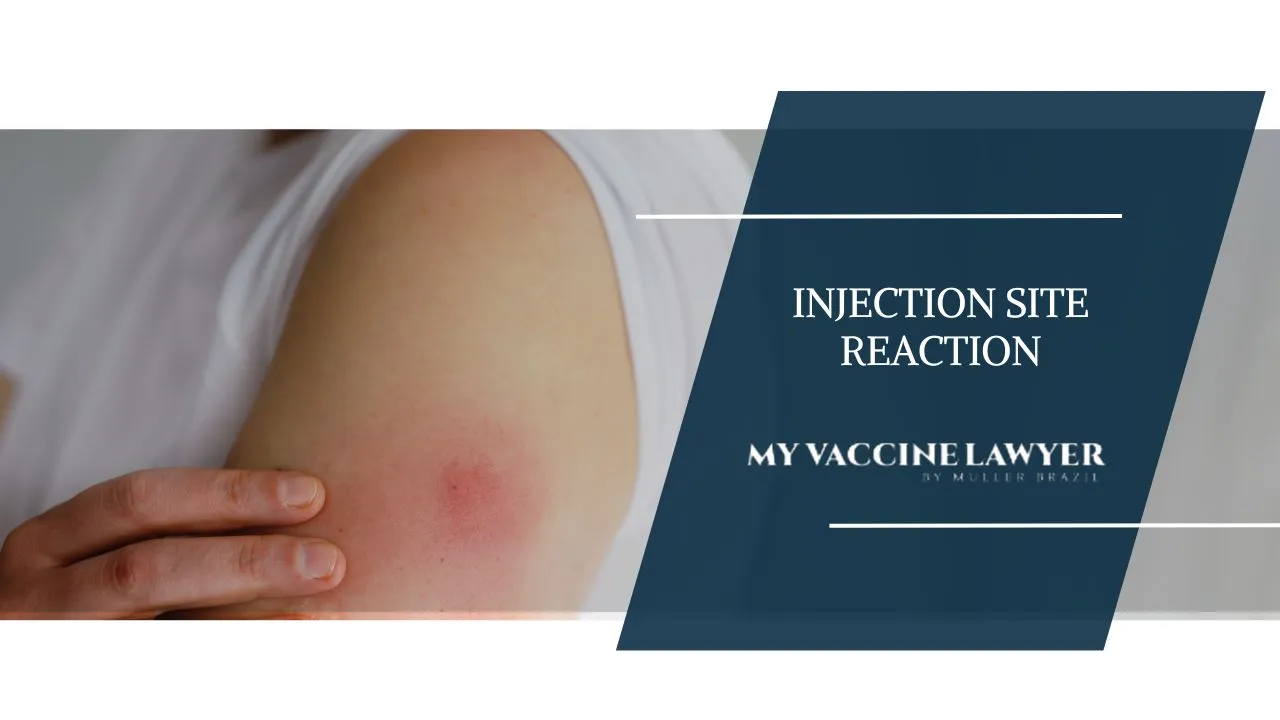
When administering injections, monitoring for potential adverse reactions, including allergic responses or inflammation, is important to protect patient safety and address any complications swiftly. Common side effects of such treatments may include pain, swelling, rash formation, bleeding on the injection site, redness, and itching.
If you see signs of an allergic reaction, you must act fast. These signs include trouble breathing, low blood pressure, swelling of the tongue or throat, widespread hives and rashes, and itching. The lips and eyes might also swell up.
To provide the safest and most effective medication delivery, healthcare professionals must understand all aspects of injections. From what types are available, their use-case scenarios, proper injection sites, and advantages and drawbacks. Knowing this information allows both patients and caregivers alike to make an informed decision concerning which type is best suited for them.
By adhering strictly to safety protocols when administering injections, we reduce potential risks or complications that may arise, thereby providing a superior experience during treatment. Keeping up with the current knowledge base on injection therapies whilst staying alert will ensure desired results each time they’re utilized properly.
Injections come in five main varieties: intravenous, intramuscular, subcutaneous, intraosseous injection, and intradermal. Each of these types is administered differently to suit the individual’s needs for medication delivery.
Intramuscular steroid injections are administered in several locations. The upper arm’s deltoid muscle, vastus lateralis (thigh), ventrogluteal (hip) and dorsogluteal muscles of the buttocks are some of them.
Injecting drugs can be administered in three ways: subcutaneous, intramuscular, or intravenous injection.
Use the aseptic technique with unopened needles and syringes for maximum sterility in an injection. This will ensure that no contamination or exposure occurs throughout the process.
Reactions to injections that could indicate allergies include difficulty breathing, hives or rash, swelling in the lips, eyes, and other parts of the body as well as itching. There may be a drop in blood pressure.
Paul Brazil is a native of Dunmore, Pennsylvania and a graduate of Dunmore High School. For his undergraduate education, he attended Bloomsburg University where he majored in political science. He then went on to earn his JD from Widener University School of Law. Following graduation from law school, Mr. Brazil worked at a large Philadelphia civil defense firm where he litigated workers’ compensation claims and Heart and Lung Act cases. In 2012, he joined with his coworker Max Muller to form Muller Brazil.
In this blog post, we will dive into the various types of vaccine injection site reactions, their symptoms, treatment and how to prevent and manage...

Injections are a routine part of medical care, used for vaccinations, medications, and other treatments. For most people, side effects are limited to...

Getting a vaccine is a routine part of staying healthy, but that doesn’t mean it’s always comfortable. If you've recently received a pneumonia shot,...